Axel Budde
Patient-Centered Outcomes Researcher & Clinical Data Scientist
Biography
Psychologist (M.Sc. Hons, University of Amsterdam) with seven years of clinical research experience across oncology, palliative care, and public health. Specialising in patient-centred outcomes, psychometric validation, and clinical data science. PhD candidate at the University of Bonn, Faculty of Medicine (Epidemiology): machine-learning prediction of quality of life after childhood cancer.
I have published on COA instrument harmonisation — linking paediatric and adult patient-reported outcome measures in an international osteosarcoma cohort (European Journal of Cancer, 2022) — and have a manuscript in preparation on non-response prediction in childhood cancer survivor surveys using machine learning.
On the applied side, I build R Shiny tools that make complex statistical methodology accessible to clinical researchers, including a psychometric analysis tool for IRT-based item fit evaluation and a dashboard developed for the Lancet Commission on Palliative Care and Pain Relief.
Download my CV.
- Patient-Centered Outcomes Research
- Psychometrics & COA Validation
- Clinical Data Science
- R & R Shiny Development
PhD Candidate in Epidemiology (expected completion: 2027)
University of Bonn, Germany
MSc in Psychology (With Honours), 2004
University of Amsterdam (UvA), The Netherlands
Diploma in Criminal Science, 1997
University of Pau and Adour Countries (UPPA), Pau, France
Skills
Recent Projects
Evaluating the practical significance of item misfit in psychological and educational tests (axelbudde.shinyapps.io/FUNForFit)
- Rasch model (1PL) and 2PL IRT analysis of weighted MNSQ fit statistics – methods central to Clinical Outcome Assessment (COA) validation
- 2D IRT modelling to estimate correlation bounds after Köhler & Hartig (2017)
- Step-guided interface with interactive result visualisation (Plotly)
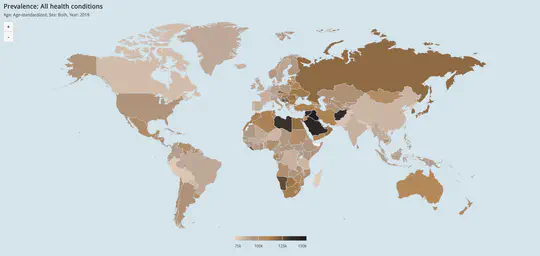
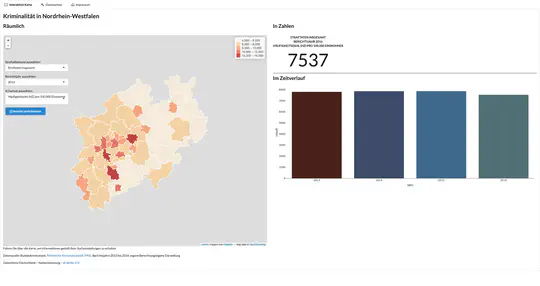
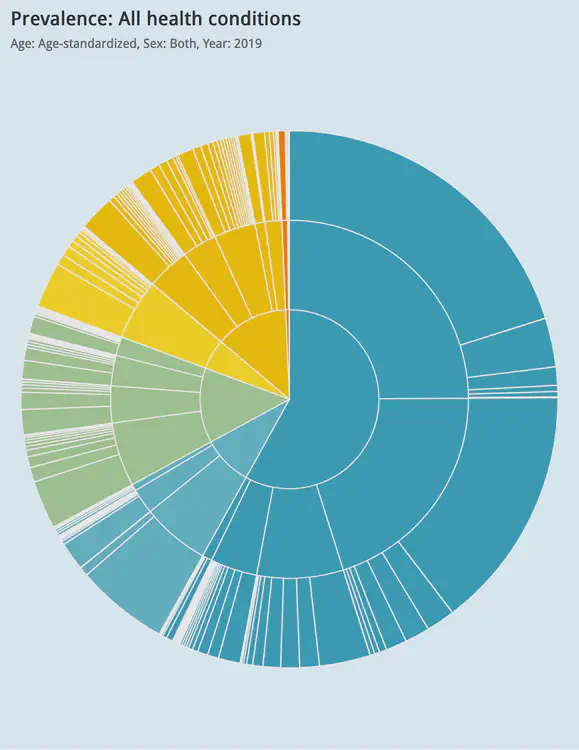
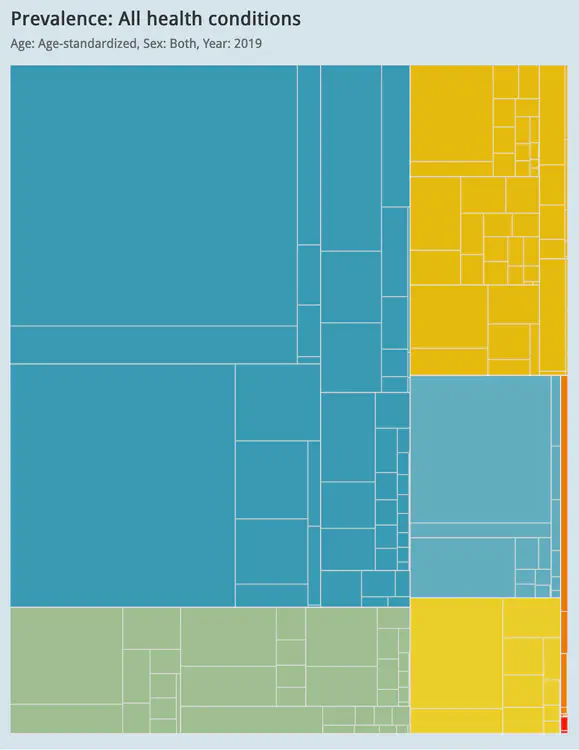
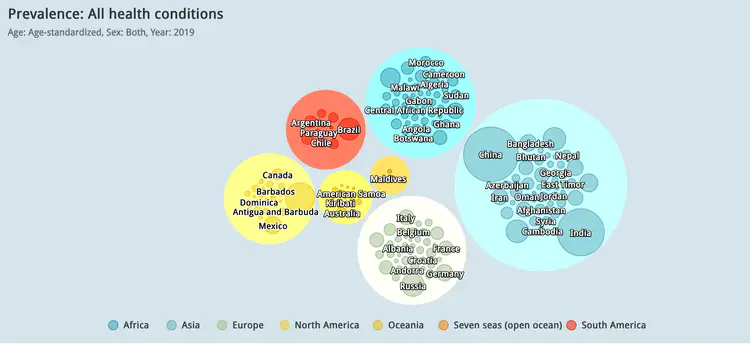
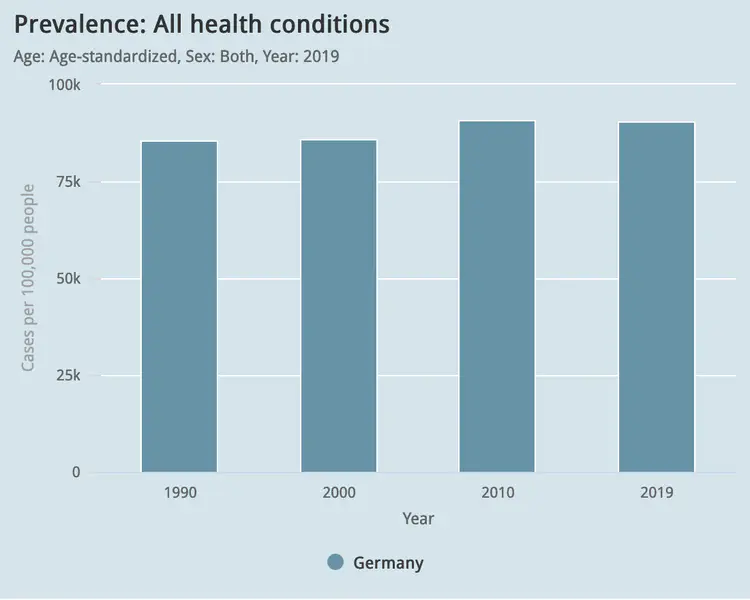
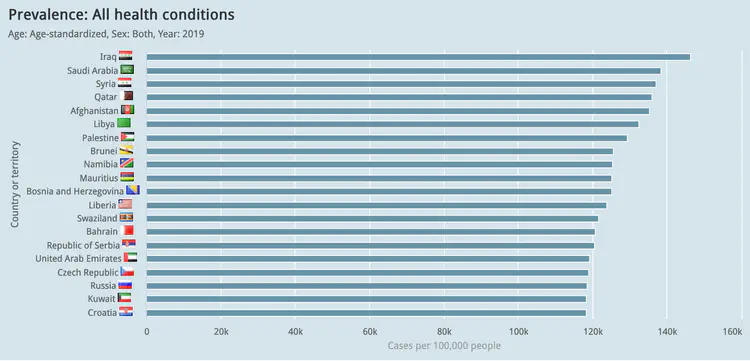
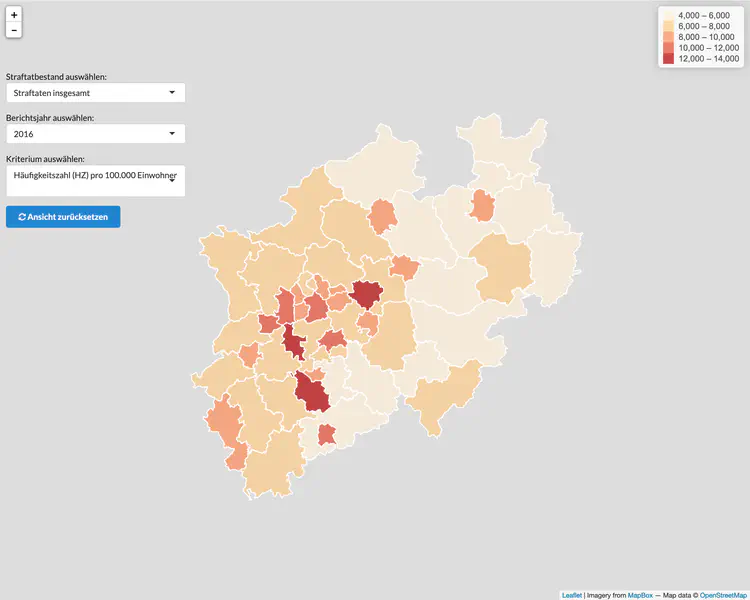
Global visualisation of Serious Health-Related Suffering (SHS) burden data (Live dashboard) – developed for the Lancet Commission on Palliative Care and Pain Relief
- Patient-centered epidemiological endpoint: SHS operationalises unmet need for palliative care across 195 countries
- Interactive maps, time series charts, and hierarchical visualisations
- Responsive web dashboard with complex filtering for research and advocacy audiences
Commercial freemium SaaS application for airline fare routing rule analysis (routingruler.com)
- Parses complex, heterogeneous rule sets from multiple industry data sources (ExpertFlyer, KVS Tool, Amadeus GDS)
- Multi-dimensional constraint logic across stopovers, transfers, IATA geographic zones, and carrier restrictions
- EgoTrip mode for instant rule-compliance verification; carrier and alliance filtering
- Full end-to-end product ownership: algorithm design, development, deployment, and commercial subscription management
Experience
Lectures in Psychology degree programme (B.Sc.):
- Introduction to psychology and its history
- Research methods
- Psychotherapy
Development of R Shiny app for visualisation of Serious Health Related Suffering (SHS):
- Developed for Lancet Commission on Palliative Care and Pain Relief
Coordination of project “E-Surv” (Funding: German Cancer Aid):
- Randomised study (n = 2,000, German Childhood Cancer Registry) comparing paper-based and eCOA administration of validated PRO instruments including EORTC-QLQ-C30, SF-36, and Multidimensional Fatigue Inventory — a direct eCOA mode equivalence study
- Logistic regression modelling of response rate predictors (sex, socioeconomic deprivation, time since diagnosis)
- Metadata harmonisation across multiple paediatric oncology studies (COSS, CESS, CWS) using Samply.MDR metadata repository
Graduate programme “Epidemiological Research Using Cancer Registry Data” (German Cancer Aid):
- July - August 2019: University of Mainz, German Childhood Cancer Registry – Comparative non-response analysis
- January - February 2019: University of Bern, Switzerland, Institute of Social and Preventive Medicine (ISPM), Paediatric Cancer Epidemiology Group (Prof. Dr. Claudia Kühni) – Development of Shiny app for space-time analyses of Swiss Childhood Cancer Registry data
Developed R Shiny apps:
- Coordination of federal-state network for online documentation system Dot.sys
- Coordination of federal-state network for relaunch PrevNet/Inforo online
- Coordination of EU project “Reducing Alcohol Related Harm” (RARHA) (Work Package Co-Lead)
- Representation within German Monitoring Centre for Drugs and Drug Addiction (DBDD) and vis-à-vis European Monitoring Centre on Drugs and Drug Addiction (EMCDDA)
Technical Skills
Programming
- R (daily since 2017 – expert)
- R Shiny (daily – expert)
- SQL (often), LaTeX (often), R Markdown (often)
Outcomes Research & Psychometrics
- Clinical Outcome Assessment (COA) instrument analysis
- Item Response Theory (IRT): Rasch / 2PL models (TAM package)
- Patient-Reported Outcome (PRO) / eCOA methodology
- Survey mode effects, non-response analysis
- Psychometric validation (fit statistics, reliability, validity)
Statistics & Data Science
- Hypothesis testing, KPI development, data cleaning
- Reproducible workflows, machine learning (random forests, penalised regression)
Documentation & Reporting
- R Markdown (expert), LaTeX (often), Git/GitHub (proficient)
Cloud & Deployment
- Render (daily), Supabase (often), AWS (often)
Additional Tools
- Metadata database MDR.Samply (often)
- SaTScan (often), QGIS (rarely)
- MySQL (often)
Languages
- English – Business fluent, IELTS (Academic) Band 8.5/9
- Dutch – Business fluent
- French – Fluent
- Spanish – Advanced basics
- German – Native
Scholarships
Projects
Featured Publications
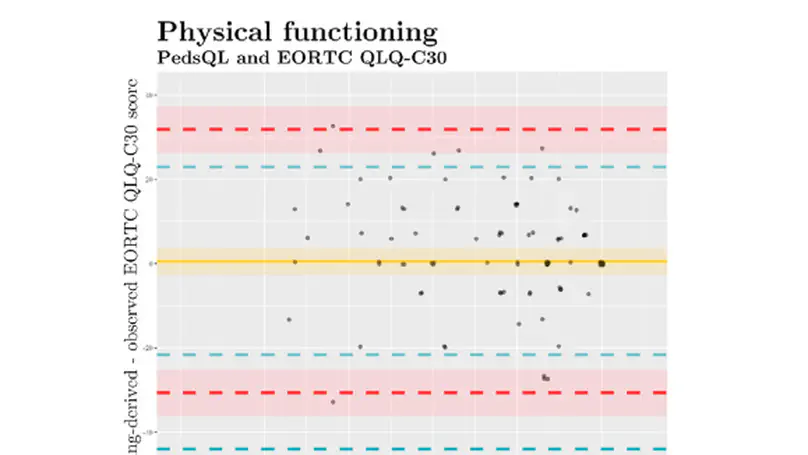
Purpose The available questionnaires for quality-of-life (QoL) assessments are age-group specific, limiting comparability and impeding longitudinal analyses. The comparability of measurements, however, is a necessary condition for gaining scientific evidence. To overcome this problem, we assessed the viability of harmonising data from paediatric and adult patient-reported outcome (PRO) measures. Method To this end, we linked physical functioning scores from the Paediatric Quality of Life Inventory (PedsQL) and the Paediatric Quality of Life Questionnaire (PEDQOL) to the European Organisation for Research and Treatment of Cancer Core Questionnaire (EORTC QLQ-C30) for adults. Samples from the EURAMOS-1 QoL sub-study of 75 (PedsQL) and 112 (PEDQOL) adolescent osteosarcoma patients were concurrently administered both paediatric and adult questionnaires on 98 (PedsQL) and 156 (PEDQOL) occasions. We identified corresponding scores using the single-group equipercentile linking method. Results Linked physical functioning scores showed sufficient concordance to the EORTC QLQ-C30: Lin’s ρ = 0.74 (PedsQL) and Lin’s ρ = 0.64 (PEDQOL). Conclusion Score linking provides clinicians and researchers with a common metric for assessing QoL with PRO measures across the entire lifespan of patients.